Colorado Newborn Blood Spot Screening (CONBSP)
General announcements
Updates to EHR for cCMV & GAMT NBS Screening
The Colorado Newborn Blood Spot Screening Program (CONBSP) is pleased to announce that the newborn screen report will include two new conditions: targeted Congenital Cytomegalovirus (cCMV) Infection and Guanidinoacetate Methyltransferase Deficiency (GAMT) in early January 2026. Please ensure the necessary changes within the electronic health record (EHR) system are made accordingly.
1. Targeted screening for congenital cytomegalovirus (cCMV) infection
cCMV is a common virus from the human herpesvirus family (HHV-5), and it is the leading non-genetic cause of childhood hearing loss. While only 10% of infected newborns show symptoms at birth (such as low birth weight, microcephaly, jaundice, and hepatosplenomegaly), most are asymptomatic. Early detection is key to timely intervention.
Report Preview
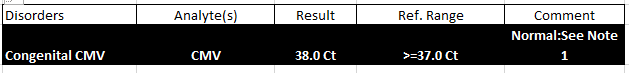
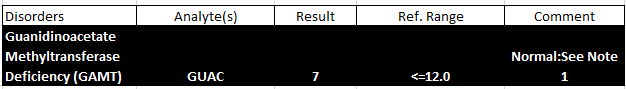
2. Guanidinoacetate Methyltransferase Deficiency (GAMT)
GAMT deficiency is an autosomal recessive condition caused by a deficiency in the GAMT enzyme, which is critical for creatine synthesis. This leads to a lack of energy for brain and muscle function.
Report Preview
NBS Results & E-Reports
Due to limited staff capacity, we kindly ask that all non-urgent newborn screening (NBS) result requests be submitted via fax at 303-691-4008 or email at cdphe_conbs@state.co.us, by completing the results request form.
To ensure timely and accurate processing, we request that inquiries for routine or non-critical results not be made by phone. Please allow up to five business days for processing these requests.
Important: Urgent cases, including those involving NICU patients, high-risk newborns, or time-sensitive situations, can still be verbally requested by phone at 303-692-3675.
If you have questions or need additional assistance, feel free to contact us at the above number or email. Please also note that our office observes quiet hours from 11:00 a.m. to 1:30 p.m., during which phone calls will go directly to secure voicemail. You are welcome to leave detailed information, and your message will be returned as soon as possible.
We appreciate your partnership and understanding as we work to provide the highest quality service. Prior to calling or faxing, check E-portal. If the report is unavailable, contact the birthing facility initially.
Sign up for an E-report account to access the portal to obtain newborn screen reports. For any issues with obtaining access or signing into the portal please email cdphe_nbs_software@state.co.us
NBS CF/NGS Go-Live Announcement
Starting on September 15, 2025, the Colorado Newborn Blood Spot Screening Program (CONBSP) will expand our Cystic Fibrosis (CF) testing by utilizing next generation sequencing (NGS). This advanced method will increase the current variant panel from 39 to 153.
What This Means for You
The name of the panel will be changed from DNA to NGS. You should also anticipate changes to the report format, which may require updates to your facility’s health information management or electronic health records systems. The number of variants identified will be included in the report; however, the specific variants themselves will not be listed. Please note that although the panel has expanded, some variants will not be detected. Abnormal results will be called out to both the primary care provider (PCP) and specialists at The CF Center at The Children’s Hospital (CHCO) in Aurora.
If you have any questions, contact Newborn Screening Follow-up, at 303-692-3675 or CDPHE_CONBS@state.co.us
Newborn Screening: New Conditions Stakeholder Input 2024/2025
The Colorado Newborn Screening Program is seeking feedback from Colorado residents, stakeholders, and interested parties regarding the appropriateness of adding new conditions to our screening panel. If this is of interest to you, we encourage you to share your input through the survey.
Transition to E-Reports
CONBSP will no longer send out physical mailers or fax the daily reports for the newborn screen to any health facilities. Reports can still be requested by phone or fax by submitting a completed results request form. Prior to calling or faxing, check E-portal. If the report is unavailable, contact the birthing facility.
Sign up for an E-report account to access the portal to obtain newborn screen reports.
If you are unable to access this document contact cdphe_nbs_software@state.co.us.
New Electronic Health Record (EHR) System Integration Underway

NCAA/Sickle Cell Requests/Parent Sample Requests
To protect your privacy, please request NCAA/Sickle Cell newborn screening results directly from the birthing facility or your child’s primary care physician. For more information, visit the Sickle Cell Screening and NCAA Rules PDF and the new sickle cell information webpage.
Health care providers and laboratories
Abnormal Results | Education | Training | Quality Improvement (QI) | E-Report | Result Request | Corrections Request
Meeting Schedule | Newsletter | Disorders | QI | Policies
Common Questions | Sickle Cell Information | Disorders
Policies
- CLIA license
- Sickle cell screening and National Collegiate Athletic Association (NCAA) rules
- NBS Retention Schedule
- HB18-1006 Newborn Screening
- Newborn screening rules and regulations
- Newborn Screening Fees for Providers
- Newborn Screening cCMV Sample Release Policy
- Please Note:
- CONBSP will accept all necessary newborn blood spot screenings for an infant up to 12 months of life.
- For refugee status, knowing the infant’s age will help with the interpretation of the results since they will be evaluated outside of the usual parameters.
Newborn Blood Spot Screening Phone: 303-692-3675 | Fax: 303-691-4008 | Email: cdphe_conbs@state.co.us